CROMSOURCE Regulatory specialists are delighted to offer a broad range of services to medical device, pharmaceutical, and biotechnology companies. We have extensive experience of regulatory document preparation together with the management of client-focused projects with a strong record of successful outcomes. We provide timely strategic advice to facilitate our clients’ product development and lifecycle maintenance helping them make their product a commercial success.
What You Need to Know About EU Clinical Trials Regulation
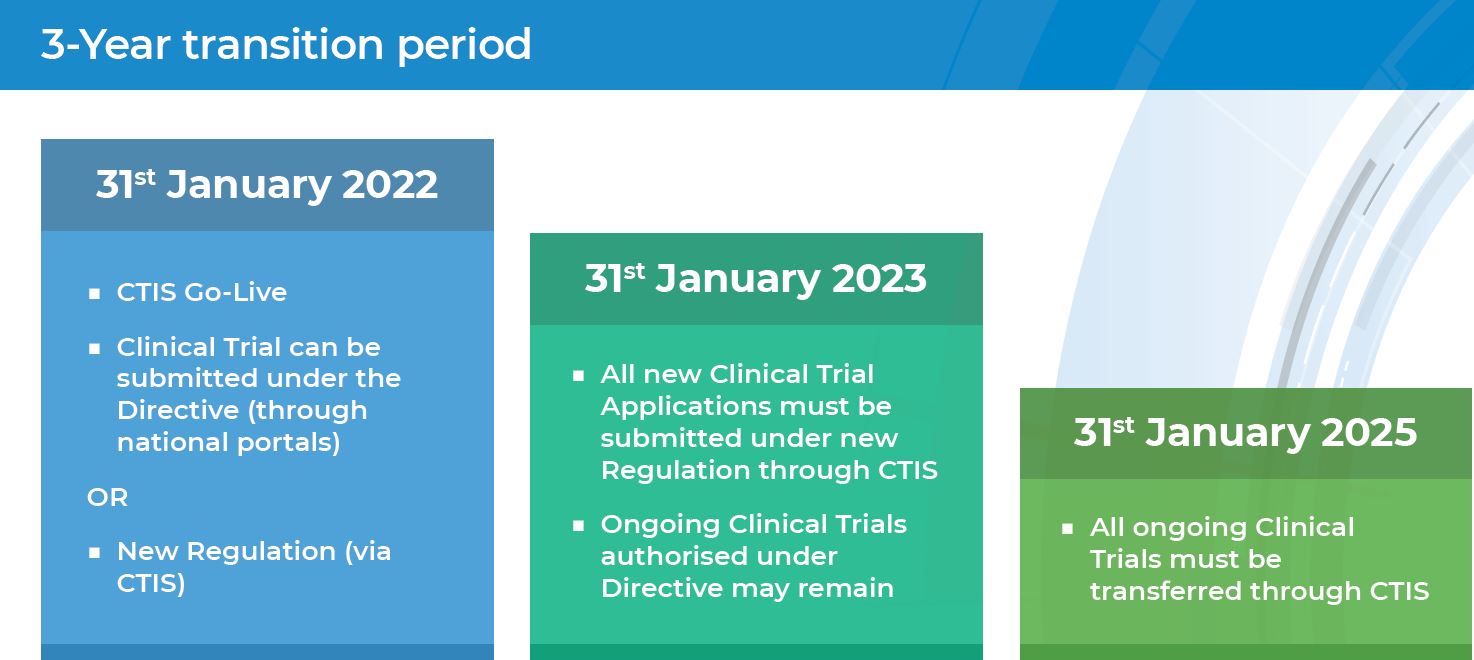
Starting from 31 January 2022 the EU Clinical Trials Regulation No 536/2014 (CTR) became applicable in all EU/ European Economic Area (EEA) Member States replacing the EU Clinical Trials Directive (2001/20/EC) (CTD). On the same day, the Clinical Trials Information System (CTIS), go-live version was opened for users to submit clinical trials on medicinal products for human use under coordination authorisation procedure.
Without a doubt, the CTR application has an impact on the clinical trials environment. Among others, the CTR harmonises the approval process for clinical trials, increases coordination between Member States, as well as maintains the highest level of standards for patient safety, and increases transparency of clinical trial information.
To learn more read our latest Factsheet “Key Insights about EU Clinical Trials Regulation” or the White Paper “The EU Clinical Trials Regulation − What You Need to Know”.
Country Specific Requirements for Clinical Projects
We maintain a Clinical Studies Submissions Database (CSSD) with comprehensive information relating to study requirements and procedures for medicinal products and medical devices in all European countries, the US, Canada and more than 15 countries worldwide. This includes information on relevant legislation/regulation, sequence of submissions to Competent Authorities and Ethics Committees, timelines, notifications, fees, and lists of documents required. Leveraging CROMSOURCE’s global presence, our regulatory intelligence professionals access, summarise, and assess global regulatory intelligence to ensure submissions are compliant with regional and local requirements.
We offer:
• Consultation on legislation and country specific requirements for clinical studies with medicinal products and medical devices.
• Liaison with National Competent Authorities, Ethics Committees, Institutional Review Boards, EU Notified Bodies, Health Canada, and the US FDA, including providing advice and preparing dossiers and applications.
• Tailored training in Regulatory Affairs – legislations, regulations, and local requirements.
Regulatory Support for All Stages of Product Lifecycle in the US and Europe
In the US, the FDA is the regulatory authority that oversees public health by assuring the safety, efficacy, and security of drugs, biological products, combination products, and medical devices. CROMSOURCE regulatory specialists in the US work in concert with clients to determine the most expeditious way to market their products for commercial success.
CROMSOURCE has a successful FDA submission track record with investigational and marketing applications for drugs and biologics. Commercial INDs are required to be submitted in the compliant eCTD format.
In Europe, the EU Member States have different National Competent Authorities (NCAs) which play an essential role in the protection of public health by assuring the quality, safety, and efficacy of medicines, Advanced Therapy Medicinal Products (ATMPs), and medical devices. CROMSOURCE regulatory experts in Europe have an established robust regulatory strategy with all NCAs to allow for accelerating start-up and study approvals.
CROMSOURCE works with Advanced Therapy Medicinal Products in the EU and US comprising a category of innovative and complex biological products , which in most cases requires extensive and complicated preclinical and clinical developments.
CROMSOURCE supports our clients through the entire classification and application processes from development, to registration and ultimately post-approval management.
Regulatory Intelligence
CROMSOURCE’s Regulatory Intelligence team:
• Detects changes and provides surveillance in the regulatory environment in the US, Europe and other countries worldwide;
• Provides timely and efficient communications with clients, supported by a comprehensive regulatory database and library.
CROMSOURCE’s Offerings
CROMSOURCE’s regulatory team provides expert guidance and advice in the following areas:
• Regulatory Newsletter to stay current on country, regional, and global clinical trial regulatory requirements and changes;
• US Agent and Regulatory Liaison for communications;
• Preparation support for FDA and Competent Authority meetings, negotiations, briefing packages, communications and rehearsals;
• Research and Commercial IND and IND maintenance;
• ANDA, NDA, BLA for US FDA;
• MAA, IMPD for Europe’s Competent Authorities;
• Clinical Trial Applications (Amendments/Notifications) for Canada and European Union countries (e.g. Voluntary Harmonisation Procedure (VHP), UK Combined Ways of Working (CWoW) and others);
• User fee payments;
• Drug establishment registration and listing.
Regulatory Consultancy
CROMSOURCE’s regulatory team provides support in the following areas:
• Strategic regulatory consulting from development to commercialisation;
• Regulatory Gap Analysis, review, and remediation of regulatory documents;
• Drug and biologic development, and regulatory pathways to market in the EU, UK, and the US;
• Smooth transitions from exploratory to confirmatory stages of investigational drugs;
• Conduct assessments of the regulatory position for new therapies and therapeutic areas;
• Post-filing support to provide complete response to regulatory inquiries;
• For all phases of product development with clients, agency meetings, and requests for scientific advice with regulatory bodies such as the EMA, FDA, MHRA, and others;
• Expedited programs, including Fast Track, Accelerated Approval, Breakthrough Therapy, and PRIME (Priority Medicines);
• Developing approval strategies in the multifaceted US 505(b)(2) drug development environment and similar programs worldwide, including Europe’s Hybrid regulatory pathway;
• Guidance and advice for optimised testing packages of nonclinical and clinical studies;
• Independent due diligence/scientific regulatory review and gap analysis of study data (CMC, nonclinical, clinical);
• Regulatory approval process of Orphan Drug Designations-Applications for rare diseases, biosimilars;
• Consultation for small molecules, biological drugs and gene/cell therapy products;
• Support clients and investigators with the preparation and submission of Institutional Review Board or Ethics Committee documentation;
• Regulatory advice for import/export permits and labeling for investigational products;
• Consultation for drug and biologic advertising, promotion, marketing and labelling;
• Regulatory submission strategy preparation and execution for multiple therapeutic areas for drugs and biologics.
CROMSOURCE Advantages
• Expert regulatory consulting team for regulatory intelligence and regulatory affairs
• Collating and analysing regulatory requirements of any product category
• Submission document management and tracking
• Regulatory content management
• Regulatory support across multiple markets and countries worldwide